-
132
Views
-
0
Comments
-
0
Like
-
Bookmark
Your brain resets and saves memories during sleep
Active systems consolidation uses sleep to turn short-term impressions into long-term memories through a dialogue between the cortex and hippocampus.
Neural coordination and the systems consolidation process
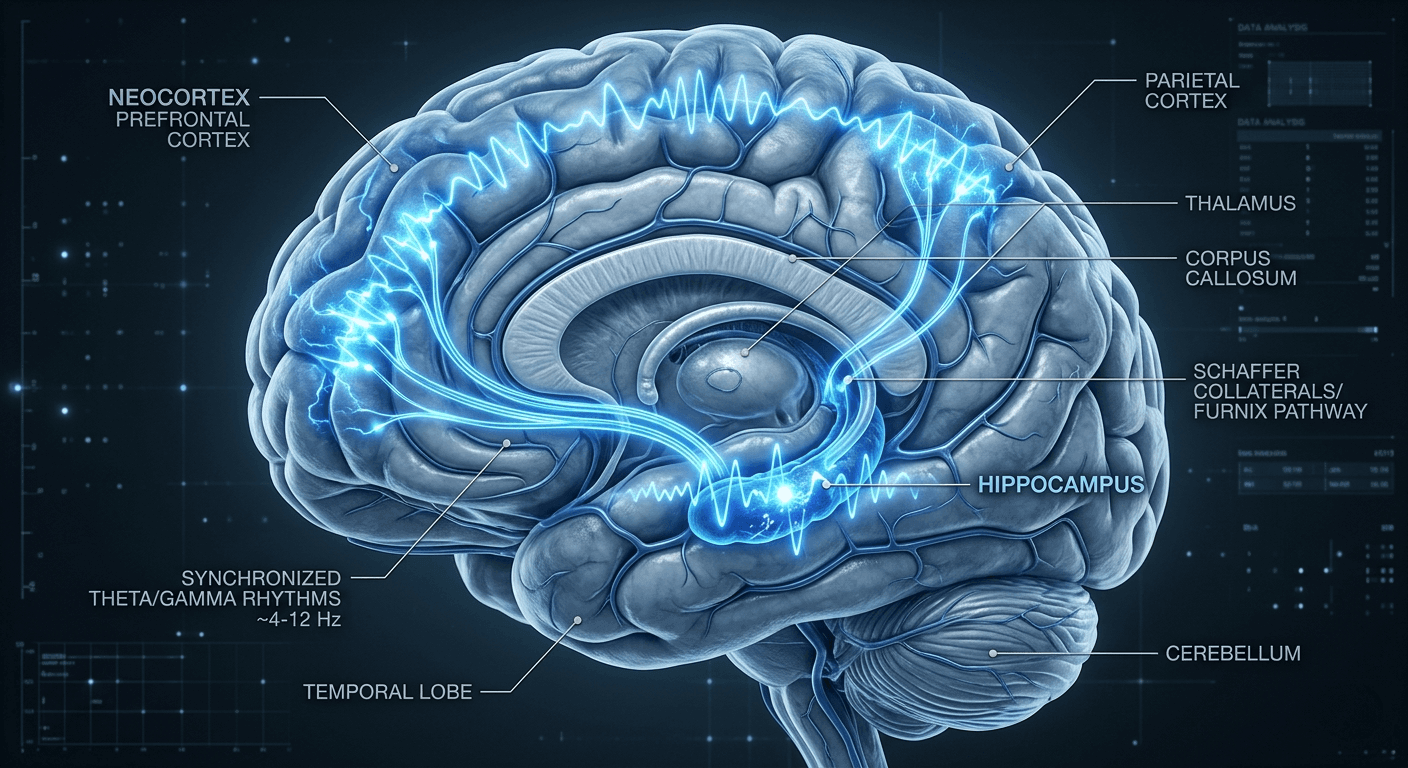
Memory is not a static preservation of experience but a dynamic biological reconstruction. This process, known as active systems consolidation, relies on the structural integrity of sleep architecture to convert volatile short-term impressions into durable long-term storage. According to neurobiological research, this transition involves a sophisticated dialogue between the hippocampus and the neocortex, characterized by repeated reactivation of memory traces alongside widespread synaptic downscaling that optimizes neural efficiency and restores plasticity for new learning.
The mechanical foundation of this transfer is neuronal replay. During non-rapid eye movement (non-REM) sleep, particularly slow-wave sleep (SWS), the brain reactivates specific firing patterns recorded during waking hours. In rodent models, hippocampal place cell ensembles re-emerge in sequential order but in a temporally compressed manner-typically by a factor of approximately 5-20 times faster than the original experience. This compression facilitates the gradual "corticalization" of memories, transitioning them from hippocampus-dependent forms into more abstract, stable, schema-like representations within neocortical networks. Human studies support coordinated reactivation during SWS following task learning, though direct quantification of compression in humans remains technically challenging.
The hierarchy of sleep stages and memory types
Sleep stages serve distinct yet complementary functional roles in information processing. Non-REM sleep, particularly deep slow-wave sleep (SWS), is the primary period for consolidating declarative memories-facts, events, and semantic knowledge. During this phase, the cerebral cortex, thalamus, and hippocampus operate in a tightly regulated loop. The cortex generates slow oscillations (typically ~0.75 Hz, often described as under 1 Hz) that act as a temporal scaffold, coordinating the activity of other brain regions through alternating up- and down-states of neuronal excitability.
Conversely, REM sleep contributes to the refinement of emotional and procedural memories, such as motor skills and emotionally salient experiences. Research indicates that REM sleep supports the modulation and selective strengthening of emotional memory traces, often linked to theta oscillations (4-8 Hz). While some studies associate REM with synaptic pruning or decorrelation of weaker connections to reduce interference and promote generalization, the precise mechanisms involve a balance of strengthening salient emotional elements and integrating or transforming representations rather than purely "pruning weaker connections" in all contexts. Theta power during REM has been positively correlated with aspects of emotional memory processing and recall, suggesting a role in emotional regulation and abstraction.
Electrophysiological markers of consolidation
The efficacy of memory stabilization depends on the precise synchronization of three key oscillations during non-REM sleep:
- Hippocampal Sharp-Wave Ripples (SWRs): High-frequency bursts (typically 80-250 Hz in rodents, ~80-100 Hz or higher in human recordings) that facilitate reactivation of latent memory traces.
- Thalamocortical Spindles: Rhythmic oscillations (10-16 Hz) that bridge communication between the hippocampus and neocortex, often nesting ripples.
- Cortical Slow Oscillations: Low-frequency waves (<1 Hz) that regulate the timing of spindles and ripples, creating windows of excitability (up-states) for effective information transfer.
These oscillations form a hierarchical nesting: slow oscillations group spindles, which in turn coordinate ripples, enabling the transfer and integration of reactivated information from hippocampal to neocortical stores. Studies show that increased ripple rates and their coupling with spindles and slow-oscillation up-states predict better post-sleep memory performance. The synaptic homeostasis hypothesis complements this by proposing that sleep oscillations help downscale synaptic strengths globally while preserving and strengthening task-relevant connections, thereby restoring metabolic efficiency and plasticity.
Representational drift and hippocampal resetting
New information is not merely stored; it is integrated and sometimes transformed through processes including representational drift. During sleep, brain activity patterns can shift, with reactivations in early non-REM periods contributing to retuning of hippocampal representations that align better with future recall or updated contexts. This evolution helps maintain flexibility in memory networks.
To sustain continuous learning capacity, the hippocampus employs resetting mechanisms. Research in rodent models demonstrates that the CA2 region can trigger periods of neuronal silence (or specific silencing events, including long barrages in certain interneurons) during deep non-REM sleep. This allows overused circuits in CA1 and CA3 to reset, preventing saturation and ensuring the same neural resources remain available for encoding new memories the following day. Such mechanisms may hold therapeutic potential for enhancing memory retention or modulating maladaptive traces, such as those in traumatic memories, though human applications remain exploratory.
External influences and efficient consolidation
Emerging research explores ways to influence these internal processes. Targeted Memory Reactivation (TMR) delivers auditory or other sensory cues during sleep to bias reactivation of specific memories, often enhancing ripple activity, spindle coupling, and consolidation when timed to slow-oscillation up-states. This demonstrates that consolidation, while largely unconscious, can respond to external stimuli under optimal electrophysiological conditions.
Furthermore, data from a 2025-2026 study on advanced meditation practitioners (published around April 2026) indicate that long-term practitioners exhibit a biological brain age approximately 5.9 years younger than chronological age, as estimated from sleep EEG patterns. These individuals show heightened high-amplitude bursts and organized activity during lighter non-REM stages (e.g., N2), linked to improved inhibitory circuits and synaptic dynamics. Such findings suggest cognitive practices like meditation may enhance sleep efficiency and restorative processes, potentially optimizing consolidation without necessarily reducing total sleep time. Additionally, even brief periods of wakeful rest (e.g., a few minutes of quiet, eyes-closed offline pause) immediately following learning can support memory stabilization by minimizing interference and allowing early offline reactivation, indicating that consolidation processes begin rapidly upon withdrawal of attention from external tasks.
Key takeaways
- Long-term memory formation involves active systems consolidation with transfer and transformation of representations from the hippocampus to neocortical networks.
- Neuronal replay during non-REM sleep, particularly SWS, reactivates waking patterns, often in temporally compressed form (roughly 5-20 times faster in rodent hippocampal place cells).
- Effective memory consolidation requires precise temporal synchronization (hierarchical nesting) of cortical slow oscillations, thalamocortical spindles, and hippocampal sharp-wave ripples.
- REM sleep contributes to the modulation, refinement, and emotional processing of memories, including procedural and emotionally salient traces, often linked to theta activity.
- CA2 region of the hippocampus supports resetting mechanisms during deep sleep via periods of neuronal silence, helping maintain capacity for new learning.
- Targeted Memory Reactivation (TMR) can bias reactivation and enhance consolidation when cues align with slow-oscillation up-states.
- Long-term advanced meditation is associated with ~5.9 years younger biological brain age based on sleep EEG, with more efficient high-amplitude bursts in lighter sleep stages.
- Brief wakeful rest (offline pauses) following learning supports early memory stabilization by reducing interference.
Sources
- Born & Wilhelm (2012)System consolidation of memory during sleephttps://pmc.ncbi.nlm.nih.gov/articles/PMC3278619/
- Brodt et al. (2023)Sleep—A brain-state serving systems memory consolidationhttps://www.sciencedirect.com/science/article/pii/S0896627323002015
- Harrington et al. (2025)Memory consolidation during sleep: a facilitator of new learning?https://pmc.ncbi.nlm.nih.gov/articles/PMC7618826/
- Cornell University News (2024)Sleep resets neurons for new memories the next dayhttps://news.cornell.edu/stories/2024/08/sleep-resets-neurons-new-memories-next-day
- Banks et al. (2025/2026)Sleep-Based Brain Age Is Reduced in Advanced Inner Engineering Meditatorshttps://link.springer.com/article/10.1007/s12671-025-02583-y
- Wamsley (2019)Memory Consolidation during Waking Resthttps://pmc.ncbi.nlm.nih.gov/articles/PMC7024394/











